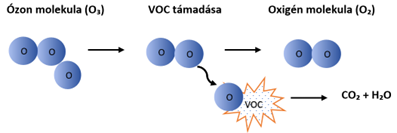
Ozone (O₃) is formed naturally from oxygen (O₂), and in our equipment it is also produced from oxygen using 185 nm UV light. Through photolysis, atomic oxygen (nascent oxygen) is formed from the diatomic oxygen molecules in the air, which then combines with the oxygen in the air to form ozone. This strongly oxidizes volatile organic compounds (VOCs), neutralizes unpleasant odors, and disinfects the air.
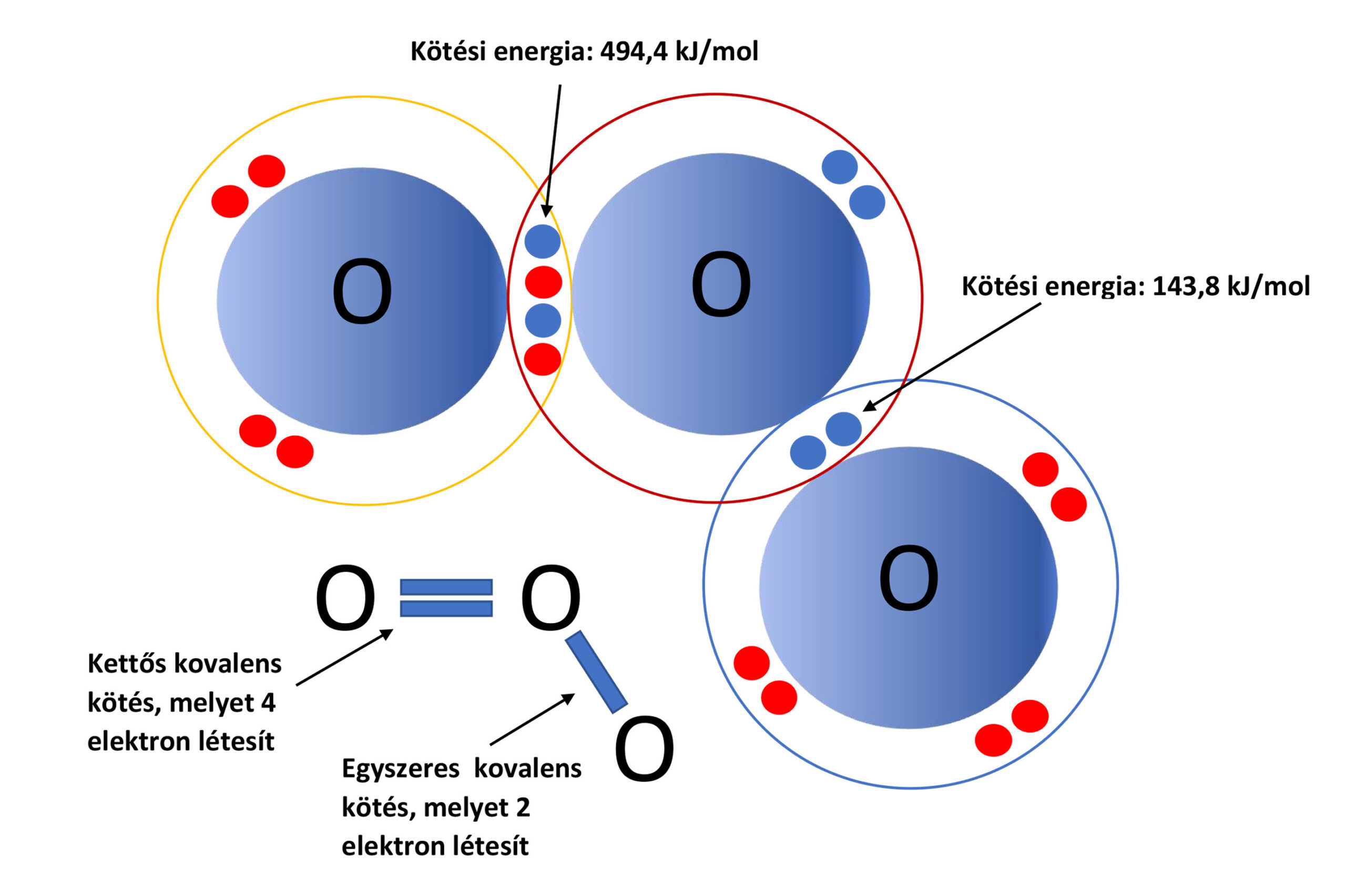
The figure above shows the electrons in the outer electron shell of the oxygen atoms in the ozone molecule. The oxygen atom, which has only a single covalent bond and a lower bond energy, is the one that easily breaks away from the ozone molecule; this is the atom we use in the kitchen exhaust duct to oxidize volatile organic compounds. Care must be taken to ensure that the ozone can travel with the airflow in the duct for at least 2–3 seconds, to allow time for it to exert its oxidizing effect.
Ozone is a "suicidal" molecule. It seeks the VOC molecule to be attacked, oxidizes it, and in the process destroys itself.
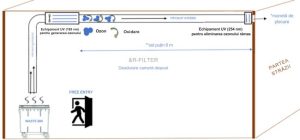
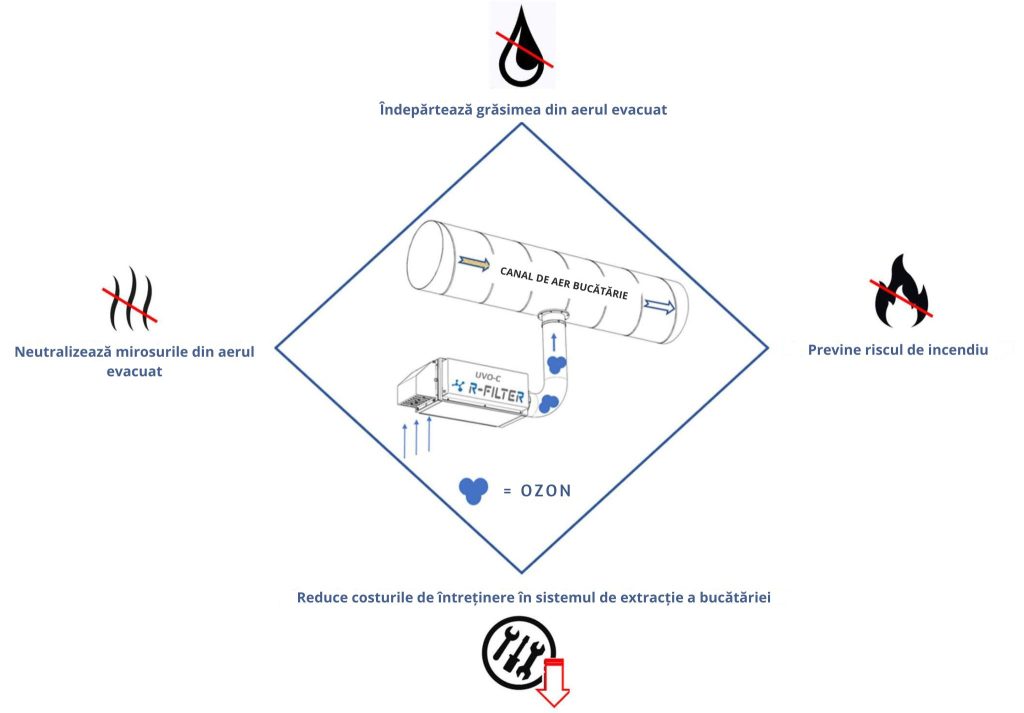
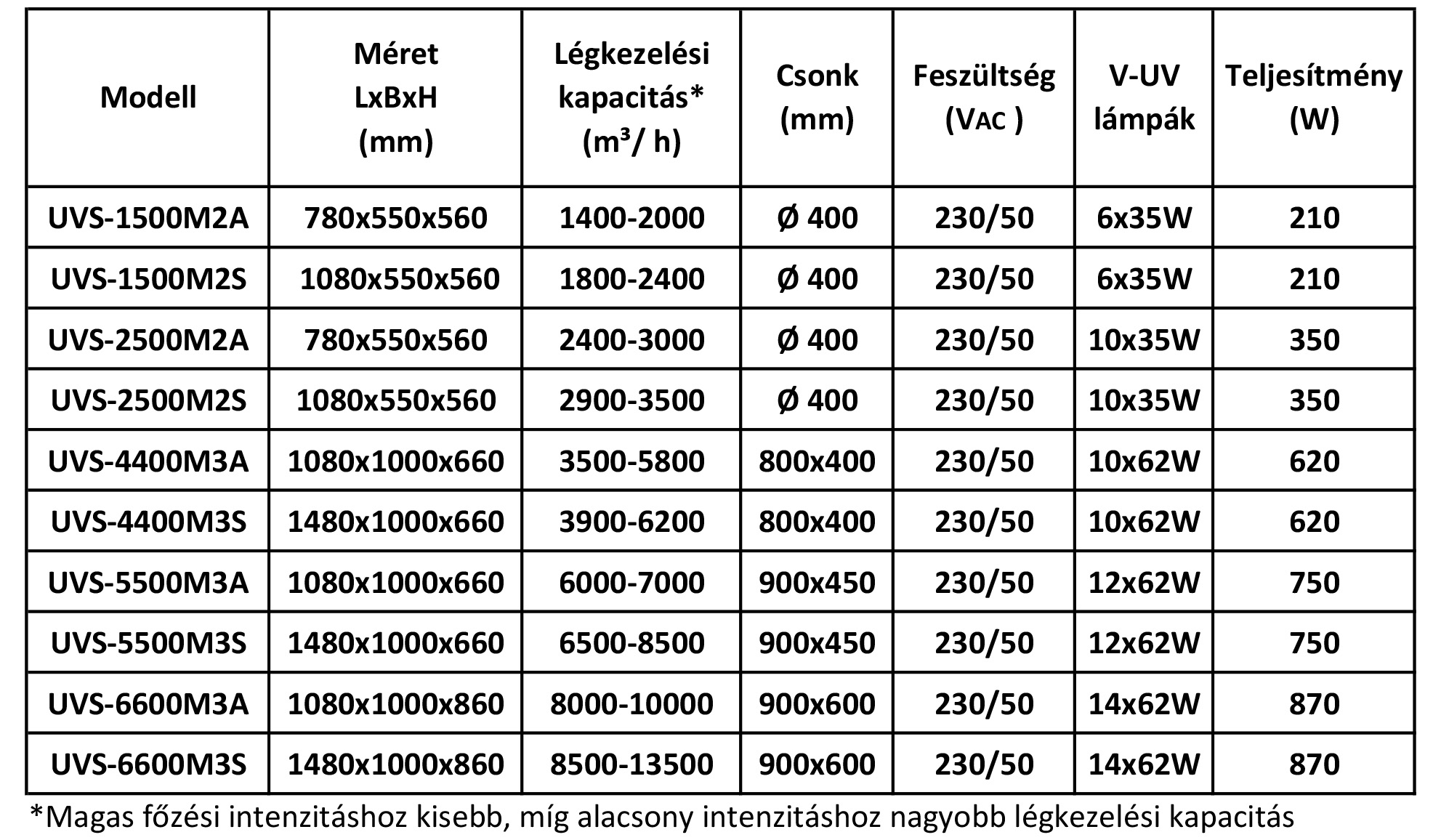
Our equipment that is installed in the ventilation duct:
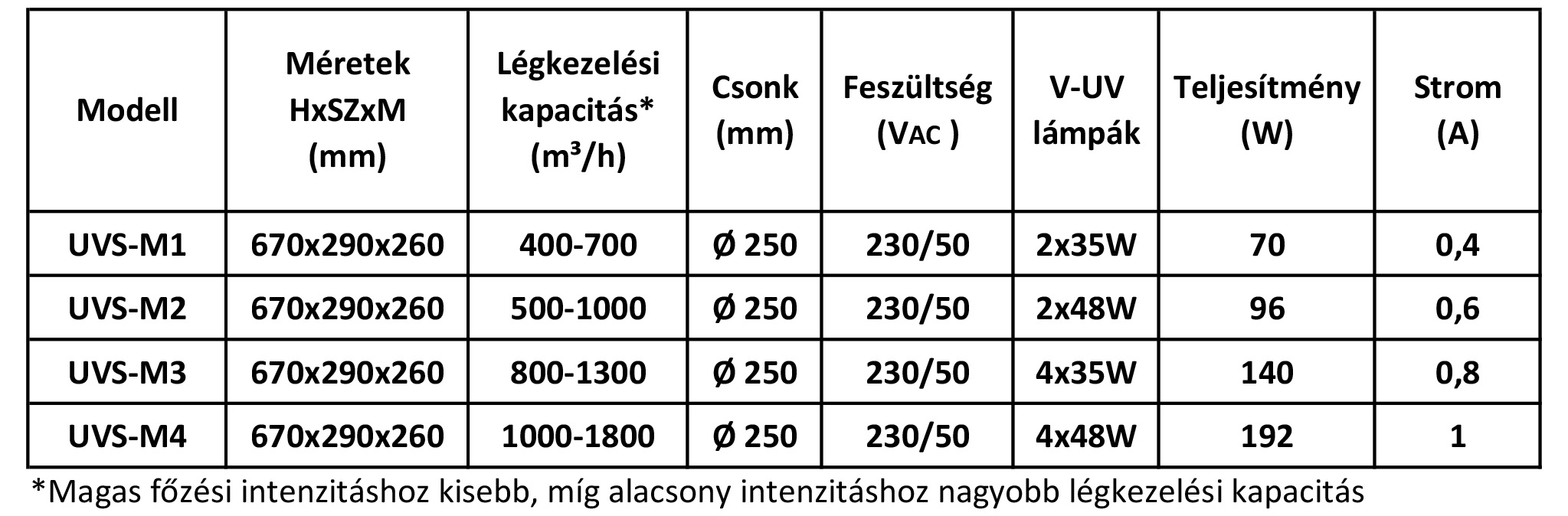
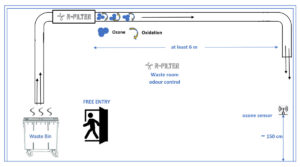
Our equipment that injects ozone into the duct from outside: