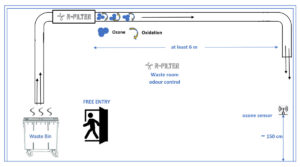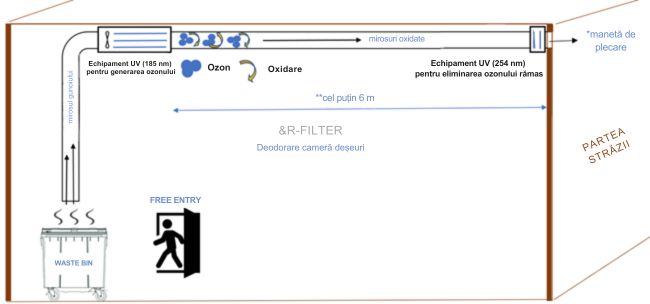
*exhaust air: If the exhaust air is directed to an enclosed area or at street level, it is important that the ozone concentration measured at the exhaust outlet does not exceed the local occupational ozone exposure limits.
**at least 6 m: Longer ducts allow the ozone to remain in contact with odors for a longer time so it can oxidize them.
Why is 254 nm UV radiation capable of decomposing ozone (O3)?
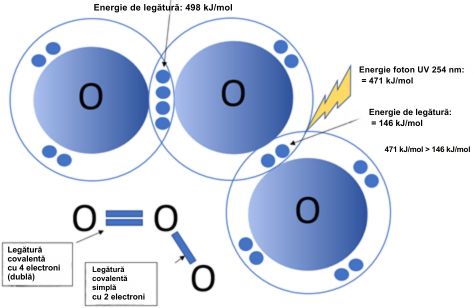
The bond energy O = O double is approximately 498 kJ/mol, and the bond energy O - O single is approximately 146 kJ/mol. The energy of a UV photon with a wavelength of 254 nm is approximately 471 kJ/mol.
The 254 nm UV radiation is capable of decomposing ozone because it has enough energy to break the bond of the ozone molecule (O₃) and decompose it into an oxygen molecule (O₂) and a free oxygen atom (O).